Sinai Health’s Dr. Jordan Lerner-Ellis
By: Jovana Drinjakovic, PhD, Scientific Communications Officer, Lunenfeld-Tanenbaum Research Institute at Sinai Health, Toronto
Sinai Health researchers have received more than $17M from federal and provincial governments for two major initiatives: creation of a national digital infrastructure for genomic data sharing and establishment of a state-of-the-art tissue engineering platform for disease research and drug discovery at the Lunenfeld-Tanenbaum Research Institute (LTRI), part of Sinai Health.
Genomic data sharing for better healthcare
In a major push to harness the power of genomic data for healthcare, the Canadian Genomic Data Commons (CGDC) initiative, co-led by Sinai Health's Dr. Jordan Lerner-Ellis, has secured over $7.4 million in federal and provincial funding.
With support from the Canadian Foundation for Innovation and the provincial governments of Alberta, Manitoba, and Ontario, the CGDC will establish a national infrastructure promoting the free flow of genomic data between clinicians, other healthcare providers and researchers.
“As genome sequencing becomes more affordable, most genomic data will be generated in the clinical setting," said Dr. Lerner-Ellis, Director of Advanced Molecular Diagnostics Laboratory at Mount Sinai Hospital and an Associate Member at LTRI.
“There is still so much we don't know about the relationship between genetics and disease and if we don't find a way to share that data for clinical and research purposes, then there's a risk of that data being lost," he said.
The CGDC will develop databases, build robust IT systems and analytical tools, and develop secure data-sharing protocols that ensure the privacy and confidentiality of patient information.
The initiative, co-led by Dr. Kym Boycott, Senior Scientist at the CHEO Research Institute in Ottawa, and a Professor of Pediatrics at the University of Ottawa, brings together hundreds of clinical geneticists and informatics experts from across the country with more than 20 clinical laboratories participating in the project.
“Genomic data hold clues to health and disease, but its interpretation remains a challenge," said Dr. Boycott, who is also Chair of Genetics and a Clinical Geneticist at CHEO.
“The CGDC is an essential research platform for sharing and interpreting genomic data, identifying mutations that lead to disease, and establishing new gene-disease associations that will ultimately help us better diagnose and care for patients and families," she said.
As well as data sharing, the team will also develop purpose-made software tools for researchers and clinicians to enable the use of the data for various applications. These tools will enable detection of disease-causing variants, discovery of new gene-disease associations and more effective treatment plans by connecting patients with clinical trials that are most relevant to their specific conditions. The CGDC will also introduce the first integrated consent platform, allowing Canadians to participate in research projects connected to their genomic data.
Read the original story.
Human in a dish
In Drs. Jeff Wrana and Laurence Pelletier's laboratories at LTRI, tiny brain-like tissues are sprouting, growing ever more complex thanks to the blood vessels supporting them.
A feat of tissue engineering, these minuscule structures, called organoids, are derived entirely from human stem cells, and may hold the clues to how the brain develops – and how it unravels in disease.
“Organoids enable us to study normal brain physiology and disorders that range from autism to bipolar disorder to Alzheimer's in a way that hasn't been possible before," said Dr. Wrana, a Senior Investigator at LTRI.
Critically, the organoids allow researchers to listen in on the crosstalk between different cells in their environment, which is important for tissue development and maintenance. When this communication is disturbed, it disrupts tissue architecture and can lead to disease.
Organoids could also be harnessed for personalized medicine by creating patient-specific tissues, enabling more precise understanding and treatment of individual health conditions.
Collaborating with Dr. Liliana Attisano at the University of Toronto, Drs. Wrana and Pelletier have created a micro-engineered 'organ-on-a-chip', which mimics the physiological environment necessary for organoid development, including the integration of blood vessels to enhance tissue maturation. Their next step will be to include immune cells that research has shown are also instrumental for proper tissue development.
Further research will be enabled by a $10 million investment from the Canadian Foundation for Innovation and the Ontario Research Fund to Drs. Wrana and Pelletier. The funding will facilitate the acquisition of state-of-the-art scientific equipment, including a single cell biology suite, high-definition microscopes, and advanced 3D bioprinters, to establish the Tissue Engineering Design Suite for the development of brain, gut, lung, kidney and other organoids. This complements LTRI's existing technology platforms that include a Canadian first – the Nikon Centre of Excellence, which is directed by Dr. Pelletier and focuses on sophisticated microscopy techniques essential for tissue engineering research.
Beyond exploring neurological conditions, the tissue engineering platform will enable the cultivation of diverse organoids, which will serve as research models for investigating a range of diseases including colorectal cancer and its metastatic processes, lung fibrosis, polycystic kidney disease, and more.
The Suite will be housed at the Network Biology Collaborative Centre (NBCC), LTRI's core research facility hub, where it can be accessed by a wider group of researchers from Sinai Health and beyond.
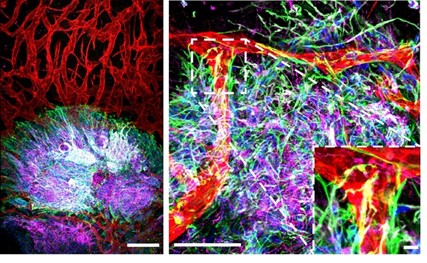
Lab-grown human brain organoid tissue derived entirely from stem cells. The left panel is a low power view, the right panel is higher power, showing integration of diverse cells types, including different kinds of brain cells, such as neurons (blue and pink) and astrocytes (green), and blood vessels (red). The inset shows a higher power view of the astrocytes making connections to the blood vessels, similar to what occurs in the body.
Read the original story.